Publications
Nature Communications (2026)
Purna Chandra Rao Vasireddy, Timothy Low-Beer, Katherine A. Spoth, Devrim Acehan, Matthew R. Crawley & Michael W. Martynowycz
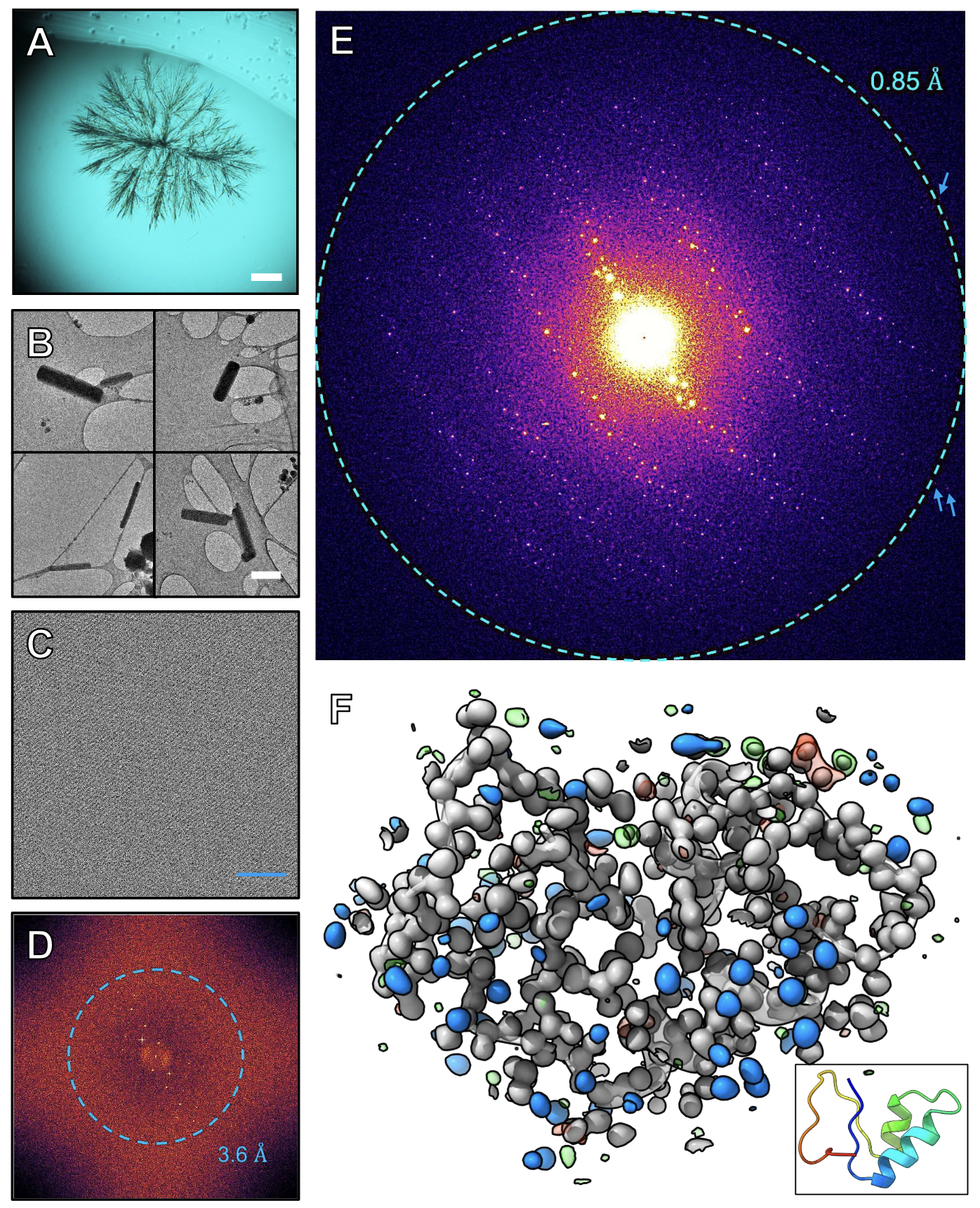
While purifying the seed protein crambin, we find that needles of pure protein nanocrystals form spontaneously during the drying of a simple ethanolic purification drop. These needles diffract X-rays weakly but are well-suited for microcrystal electron diffraction (MicroED). Merging data from 58 such nanocrystals yields diffraction to 0.85 Å resolution and solves the structure ab initio using a five-residue helical fragment to initiate density modification. The resulting map enables automated model building and resolves individual hydrogen atoms. This work reports an atomic resolution MicroED structure of the protein crambin solved ab initio. This study establishes a publicly available benchmark showing that sub-ångström ab initio MicroED can be achieved on standard 200 kV instrumentation without energy filtering or FIB milling, when serial merging is combined with anisotropy aware truncation to address preferred orientation. For this dataset, isotropic truncation alone did not produce a fragment placement suitable for phasing in this workflow, whereas applying anisotropy correction supported an ab initio solution.
Chemical Communications (2026)
Viet Thuc Dang, Michael W. Martynowycz, Dan McElhenya, Andy I. Nguyen
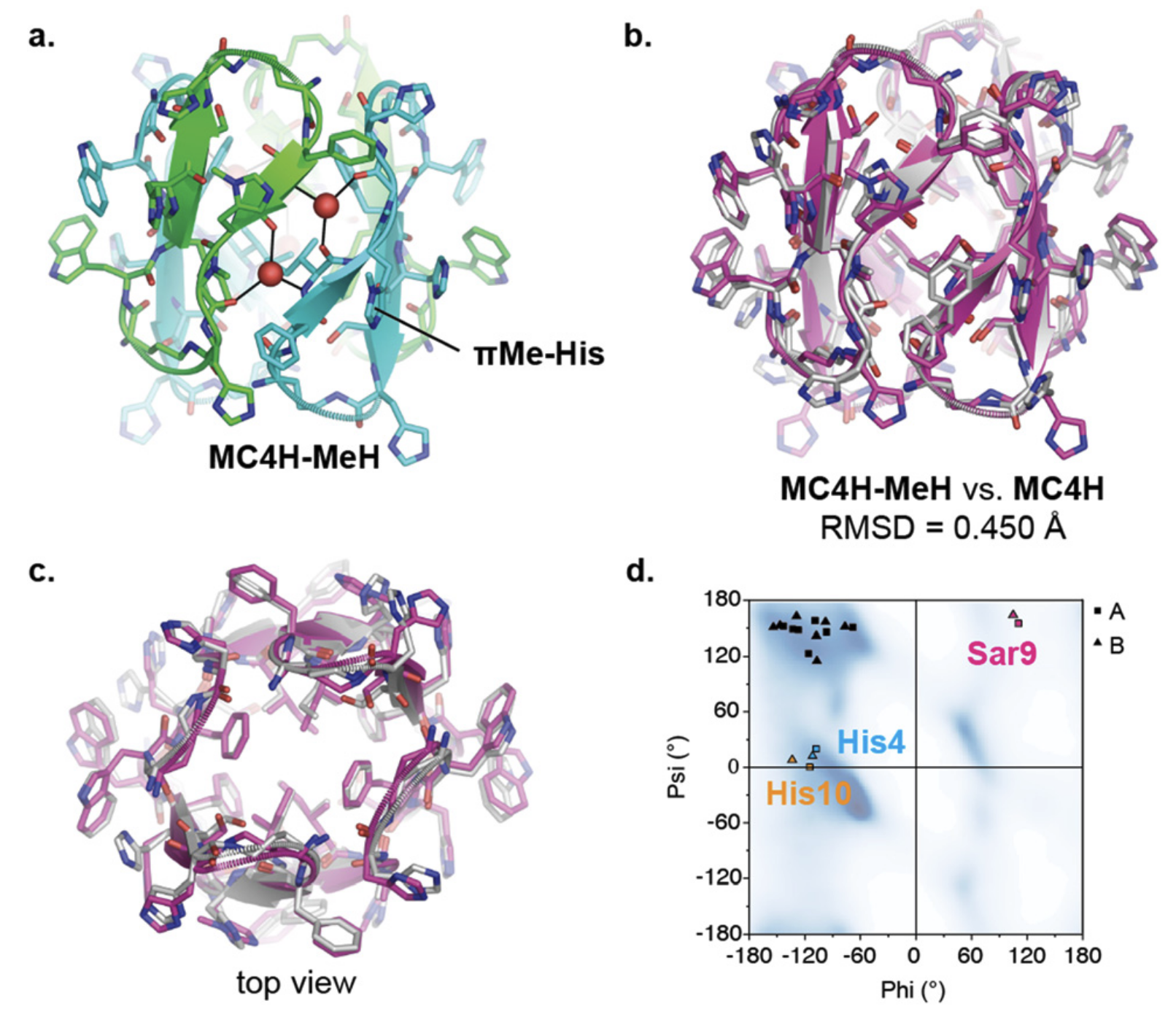
β-Barrels are ubiquitous motifs in protein structures, but the fundamental rules underlying their formation are unclear, and their de novo design remains highly challenging. Small peptides that form barrels are especially scarce. Here, we report barrels with the shortest staves (6 residues, ∼60% of previous record) and smallest shear number (S = 4) so far, formed from 12-residue macrocyclic peptides. The miniature barrel has anomalous structural features, demonstrated by solution phase and crystallographic characterization; there is a pronounced and essential backbone kink imparted by an achiral residue, N-methylglycine, as well as four structural water molecules stitching the seams of the barrel. These results provide insights into how extremely short sequences could form barrel assemblies.
Nature Communications (2025)
William J Nicolas, Anna Shiriaeva, Michael W Martynowycz, Angus C Grey, Yasmeen N Ruma, Paul J DonaldSon, Tamir Gonen
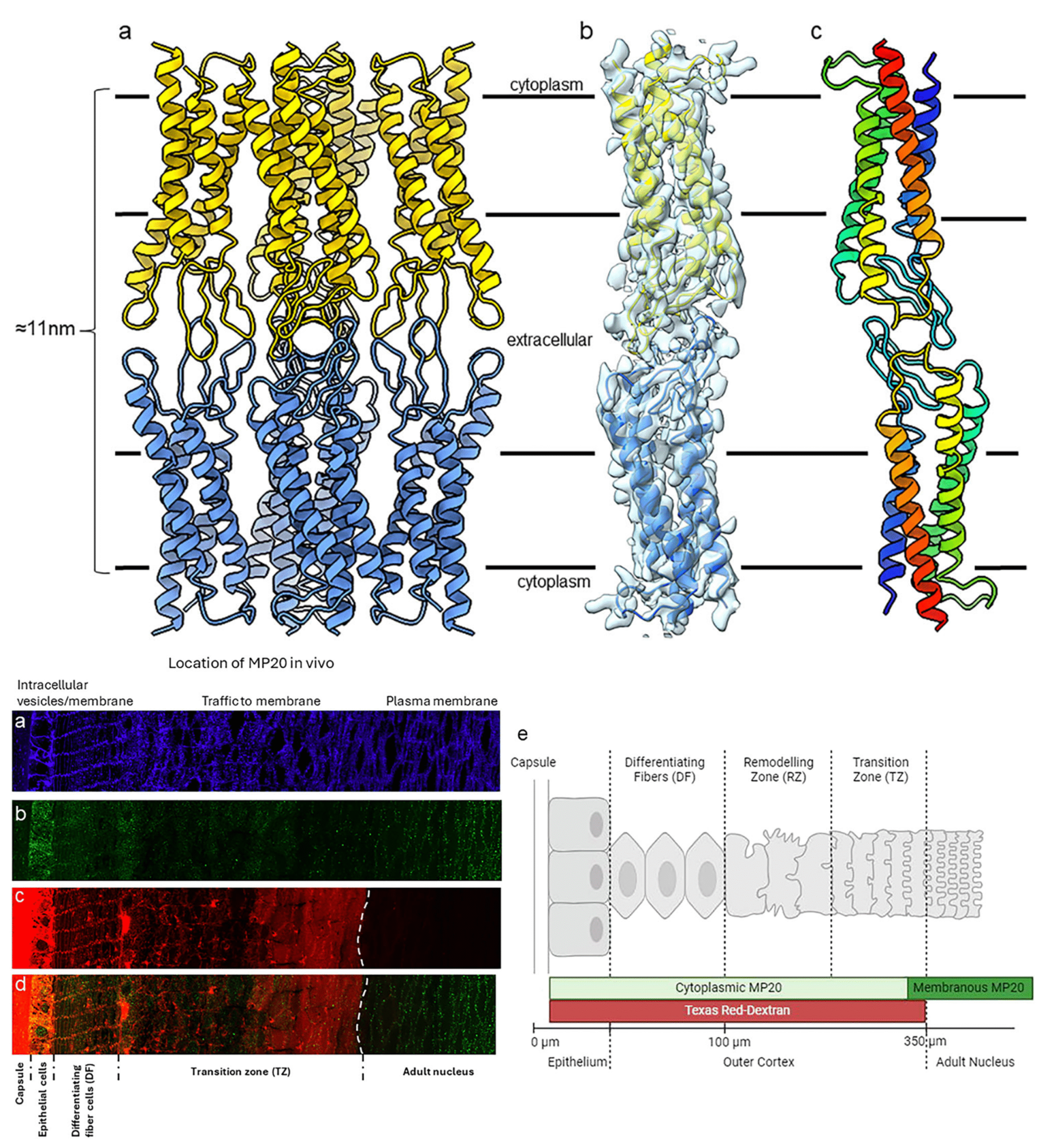
Human lens fiber membrane intrinsic protein MP20 is the second most abundant membrane protein of the human eye lens. Despite decades of effort its structure and function remained elusive. Here, we determined the MicroED structure of full-length human MP20 in lipidic-cubic phase to a resolution of 3.5 Å. MP20 forms tetramers each of which contain 4 transmembrane α-helices that are packed against one another forming a helical bundle. We find that each MP20 tetramer formed adhesive interactions with an opposing tetramer in a head-to-head fashion. Investigation of MP20 localization in human lenses indicate that in young fiber cells MP20 is initially localized to the cytoplasm in differentiating fiber cells but upon fiber cell maturation is inserted into the plasma membrane, correlating with the restriction of the diffusion of extracellular tracers into the lens. Together these results suggest that MP20 forms lens thin junctions in vivo, confirming its role as a structural protein in the human eye lens essential for its optical transparency.
Nature Communications (2025)
Max TB Clabbers, Johan Hattne, Michael W Martynowycz, Tamir Gonen
High-resolution information is important for accurate structure modeling but is challenging to attain in macromolecular crystallography due to the rapid fading of diffracted intensities at increasing resolution. While direct electron detection essentially eliminates the read-out noise during MicroED data collection, other sources of noise remain and limit the measurement of faint high-resolution reflections. Inelastic scattering significantly contributes to noise, raising background levels and broadening diffraction peaks. We demonstrate a substantial improvement in signal-to-noise ratio by using energy filtering to remove inelastically scattered electrons. This strategy results in sub-atomic resolution MicroED data from proteinase K crystals, enabling the visualization of detailed structural features. Interestingly, reducing the noise further reveals diffuse scattering that may hold additional structural information. Our findings suggest that combining energy filtering and direct detection provides more accurate measurements at higher resolution, facilitating precise model refinement and improved insights into protein structure and function.
Nature Protocols (2024)
William J Nicolas, Cody Gillman, Sara J Weaver, Max TB Clabbers, Anna Shiriaeva, Ampon Sae Her, Michael W Martynowycz, Tamir Gonen
Microcrystal electron diffraction (MicroED) has advanced structural methods across a range of sample types, from small molecules to proteins. This cryogenic electron microscopy (cryo-EM) technique involves the continuous rotation of small 3D crystals in the electron beam, while a high-speed camera captures diffraction data in the form of a movie. The crystal structure is subsequently determined by using established X-ray crystallographic software. MicroED is a technique still under development, and hands-on expertise in sample preparation, data acquisition and processing is not always readily accessible. This comprehensive guide on MicroED sample preparation addresses commonly used methods for various sample categories, including room temperature solid-state small molecules and soluble and membrane protein crystals. Beyond detailing the steps of sample preparation for new users, and because every crystal requires unique growth and sample-preparation conditions, this resource provides instructions and optimization strategies for MicroED sample preparation. The protocol is suitable for users with expertise in biochemistry, crystallography, general cryo-EM and crystallography data processing. MicroED experiments, from sample vitrification to final structure, can take anywhere from one workday to multiple weeks, especially when cryogenic focused ion beam milling is involved.


Nature Communications (2024)
Xiaoyu Liu, Xian Xia, Michael W Martynowycz, Tamir Gonen, Z Hong Zhou
Virus-induced cellular condensates, or viral factories, are poorly understood high-density phases where replication of many viruses occurs. Here, by cryogenic electron tomography (cryoET) of focused ion beam (FIB) milling-produced lamellae of mammalian reovirus (MRV)-infected cells, we visualized the molecular organization and interplay (i.e., “molecular sociology”) of host and virus in 3D at two time points post-infection, enabling a detailed description of these condensates and a mechanistic understanding of MRV replication within them. Expanding over time, the condensate fashions host ribosomes at its periphery, and host microtubules, lipid membranes, and viral molecules in its interior, forming a 3D architecture that supports the dynamic processes of viral genome replication and capsid assembly. A total of six MRV assembly intermediates are identified inside the condensate: star core, empty and genome-containing cores, empty and full virions, and outer shell particle. Except for star core, these intermediates are visualized at atomic resolution by cryogenic electron microscopy (cryoEM) of cellular extracts. The temporal sequence and spatial rearrangement among these viral intermediates choreograph the viral life cycle within the condensates. Together, the molecular sociology of MRV-induced cellular condensate highlights the functional advantage of transient enrichment of molecules at the right location and time for viral replication.
Cell (2024)
Xian Xia, Po-Yu Sung, Michael W Martynowycz, Tamir Gonen, Polly Roy, Z Hong Zhou
Unlike those of double-stranded DNA (dsDNA), single-stranded DNA (ssDNA), and ssRNA viruses, the mechanism of genome packaging of dsRNA viruses is poorly understood. Here, we combined the techniques of high-resolution cryoelectron microscopy (cryo-EM), cellular cryoelectron tomography (cryo-ET), and structure-guided mutagenesis to investigate genome packaging and capsid assembly of bluetongue virus (BTV), a member of the Reoviridae family of dsRNA viruses. A total of eleven assembly states of BTV capsid were captured, with resolutions up to 2.8 Å , with most visualized in the host cytoplasm. ATPase VP6 was found underneath the vertices of capsid shell protein VP3 as an RNA-harboring pentamer, facilitating RNA packaging. RNA packaging expands the VP3 shell, which then engages middle- and outer-layer proteins to generate infectious virions. These revealed “duality” characteristics of the BTV assembly mechanism reconcile previous contradictory co-assembly and core-filling models and provide insights into the mysterious RNA packaging and capsid assembly of Reoviridae members and beyond.


Ultramicroscopy (2024)
Andrey A Bardin, Alison Haymaker, Fateme Banihashemi, Jerry YS Lin, Michael W Martynowycz, Brent L Nannenga
We report new advancements in the determination and high-resolution structural analysis of beam-sensitive metal organic frameworks (MOFs) using microcrystal electron diffraction (MicroED) coupled with focused ion beam milling at cryogenic temperatures (cryo-FIB). A microcrystal of the beam-sensitive MOF, ZIF-8, was ion-beam milled in a thin lamella approximately 150 nm thick. MicroED data were collected from this thin lamella using an energy filter and a direct electron detector operating in counting mode. Using this approach, we achieved a greatly improved resolution of 0.59 Å with a minimal total exposure of only 0.64 e-/A2. These innovations not only improve model statistics but also further demonstrate that ion-beam milling is compatible with beam-sensitive materials, augmenting the capabilities of electron diffraction in MOF research.
Structure (2023)
Johan Hattne, Max TB Clabbers, Michael W Martynowycz, Tamir Gonen
The combination of high sensitivity and rapid readout makes it possible for electron-counting detectors to record cryogenic electron microscopy data faster and more accurately without increasing the number of electrons used for data collection. This is especially useful for MicroED of macromolecular crystals where the strength of the diffracted signal at high resolution is comparable to the surrounding background. The ability to decrease fluence also alleviates concerns about radiation damage which limits the information that can be recovered from a diffraction measurement. The major concern with electron-counting direct detectors lies at the low end of the resolution spectrum: their limited linear range makes strong low-resolution reflections susceptible to coincidence loss and careful data collection is required to avoid compromising data quality. Nevertheless, these cameras are increasingly deployed in cryo-EM facilities, and several have been successfully used for MicroED. Provided coincidence loss can be minimized, electron-counting detectors bring high potential rewards.


Structure (2023)
Alison Haymaker, Andrey A. Bardin, Tamir Gonen, Michael W. Martynowycz, Brent L. Nannenga
Microcrystal electron diffraction (MicroED) is a powerful tool for determining high-resolution structures of microcrystals from a diverse array of biomolecular, chemical, and material samples. In this study, we apply MicroED to DNA crystals, which have not been previously analyzed using this technique. We utilized the d(CGCGCG)2 DNA duplex as a model sample and employed cryo-FIB milling to create thin lamella for diffraction data collection. The MicroED data collection and subsequent processing resulted in a 1.10 Å resolution structure of the d(CGCGCG)2 DNA, demonstrating the successful application of cryo-FIB milling and MicroED to the investigation of nucleic acid crystals.
Langmuir (2023)
Michael W. Martynowycz, Konstantin Andreev, Amram Mor, David Gidalevitz
Aberrant levels of glycolipids expressed on cellular surfaces are characteristic of different types of cancers. The oligomer of acylated lysine (OAK) mimicking antimicrobial peptides displays in vitro activity against human and murine melanoma cell lines with upregulated GD3 and GM3 gangliosides. Herein, we demonstrate the capability of OAK to intercalate into the sialo-oligosaccharides of DPPC/GD3 and DPPC/GM3 lipid monolayers using X-ray scattering. The lack of insertion into monolayers containing phosphatidylserine suggests that the mechanism of action by OAKs against glycosylated lipid membranes is not merely driven by charge effects. The fluorescence microscopy data demonstrates the membrane-lytic activity of OAK. Understanding the molecular basis for selectivity toward GD3 and GM3 gangliosides by antimicrobial lipopeptides will contribute to the development of novel therapies to cure melanoma and other malignancies.


IUCRj (2023)
Cody Gillman, William J. Nicolas, Michael W. Martynowycz, Tamir Gonen
In this work, a novel crystal growth method termed suspended drop crystallization has been developed. Unlike traditional methods, this technique involves mixing protein and precipitant directly on an grid without any additional support layers. The grid is then suspended within a crystallization chamber designed in-house, allowing for vapor diffusion to occur from both sides of the drop. A UV-transparent window above and below the grid enables the monitoring of crystal growth via light, UV or fluorescence microscopy. Once crystals have formed, the grid can be removed and utilized for X-ray crystallography or microcrystal electron diffraction (MicroED) directly without having to manipulate the crystals. To demonstrate the efficacy of this method, crystals of the enzyme proteinase K were grown and its structure was determined by MicroED following focused ion beam/scanning milling to render the sample thin enough for cryoEM. Suspended drop crystallization overcomes many of the challenges associated with sample preparation, providing an alternative workflow for crystals embedded in viscous media, sensitive to mechanical stress and/or subject to on grids.
Nature Communications (2023)
Michael W. Martynowycz, Anna Shiriaeva, Max T. B. Clabbers, William J. Nicolas, Sara J. Weaver, Johan Hattne, Tamir Gonen
Crystallizing G protein-coupled receptors (GPCRs) in lipidic cubic phase (LCP) often yields crystals suited for the cryogenic electron microscopy (cryoEM) method microcrystal electron diffraction (MicroED). However, sample preparation is challenging. Embedded crystals cannot be targeted topologically. Here, we use an integrated fluorescence light microscope (iFLM) inside of a focused ion beam and scanning electron microscope (FIB-SEM) to identify fluorescently labeled GPCR crystals. Crystals are targeted using the iFLM and LCP is milled using a plasma focused ion beam (pFIB). The optimal ion source for preparing biological lamellae is identified using standard crystals of proteinase K. Lamellae prepared using either argon or xenon produced the highest quality data and structures. MicroED data are collected from the milled lamellae and the structures are determined. This study outlines a robust approach to identify and mill membrane protein crystals for MicroED and demonstrates plasma ion-beam milling is a powerful tool for preparing biological lamellae.


Journal of Structural Biology (2022)
Max T.B. Clabbers, Michael W. Martynowycz, Johan Hattne, Brent L. Nannenga, Tamir Gonen
Microcrystal electron diffraction (MicroED) uses electron cryo-microscopy (cryo-EM) to collect diffraction data from small crystals during continuous rotation of the sample. As a result of advances in hardware as well as methods development, the data quality has continuously improved over the past decade, to the point where even macromolecular structures can be determined ab initio. Detectors suitable for electron diffraction should ideally have fast readout to record data in movie mode, and high sensitivity at low exposure rates to accurately report the intensities. Direct electron detectors are commonly used in cryo-EM imaging for their sensitivity and speed, but despite their availability are generally not used in diffraction. Primary concerns with diffraction experiments are the dynamic range and coincidence loss, which will corrupt the measurement if the flux exceeds the count rate of the detector. Here, we describe instrument setup and low-exposure MicroED data collection in electron-counting mode using K2 and K3 direct electron detectors and show that the integrated intensities can be effectively used to solve structures of two macromolecules between 1.2 Å and 2.8 Å resolution. Even though a beam stop was not used with the K3 studies we did not observe damage to the camera. As these cameras are already available in many cryo-EM facilities, this provides opportunities for users who do not have access to dedicated facilities for MicroED.
Biophysical Journal (2022)
Fazle R Dayeen, Bret A Brandner, Michael W Martynowycz, Kamil Kucuk, Michael J Foody, Wei Bu, Stephen B Hall, David Gidalevitz
Cholesterol induces faster collapse by compressed films of pulmonary surfactant. Because collapse prevents films from reaching the high surface pressures achieved in the alveolus, most therapeutic surfactants remove or omit cholesterol. The studies here determined the structural changes by which cholesterol causes faster collapse by films of dipalmitoyl phosphatidylcholine, used as a simple model for the functional alveolar film. Measurements of isobaric collapse, with surface pressure held constant at 52 mN/m, showed that cholesterol had little effect until the mol fraction of cholesterol, Xchol, exceeded 0.20. Structural measurements of grazing incidence X-ray diffraction at ambient laboratory temperatures and a surface pressure of 44 mN/m, just below the onset of collapse, showed that the major structural change in an ordered phase occurred at lower Xchol. A centered rectangular unit cell with tilted chains converted to an untilted hexagonal structure over the range of Xchol = 0.0–0.1. For Xchol = 0.1–0.4, the ordered structure was nearly invariant; the hexagonal unit cell persisted, and the spacing of the chains was essentially unchanged. That invariance strongly suggests that above Xchol = 0.1, cholesterol partitions into a disordered phase, which coexists with the ordered domains. The phase rule requires that for a binary film with coexisting phases, the stoichiometries of the ordered and disordered regions must remain constant. Added cholesterol must increase the area of the disordered phase at the expense of the ordered regions. X-ray scattering from dipalmitoyl phosphatidylcholine/cholesterol fit with that prediction. The data also show a progressive decrease in the size of crystalline domains. Our results suggest that cholesterol promotes adsorption not by altering the unit cell of the ordered phase but by decreasing both its total area and the size of individual crystallites.


Nature Methods (2022)
Michael W. Martynowycz, Max T.B. Clabbers, Johan Hattne, Tamir Gonen
Structures of two globular proteins were determined ab initio using microcrystal electron diffraction (MicroED) data that were collected on a direct electron detector in counting mode. Microcrystals were identified using a scanning electron microscope (SEM) and thinned with a focused ion beam (FIB) to produce crystalline lamellae of ideal thickness. Continuous-rotation data were collected using an ultra-low exposure rate to enable electron counting in diffraction. For the first sample, triclinic lysozyme extending to a resolution of 0.87 Å, an ideal helical fragment of only three alanine residues provided initial phases. These phases were improved using density modification, allowing the entire atomic structure to be built automatically. A similar approach was successful on a second macromolecular sample, proteinase K, which is much larger and diffracted to a resolution of 1.5 Å. These results demonstrate that macromolecules can be determined to sub-ångström resolution by MicroED and that ab initio phasing can be successfully applied to counting data.
Journal of Structural Biology: X (2022)
Max T.B. Clabbers, Michael W. Martynowycz, Johan Hattne, Tamir Gonen
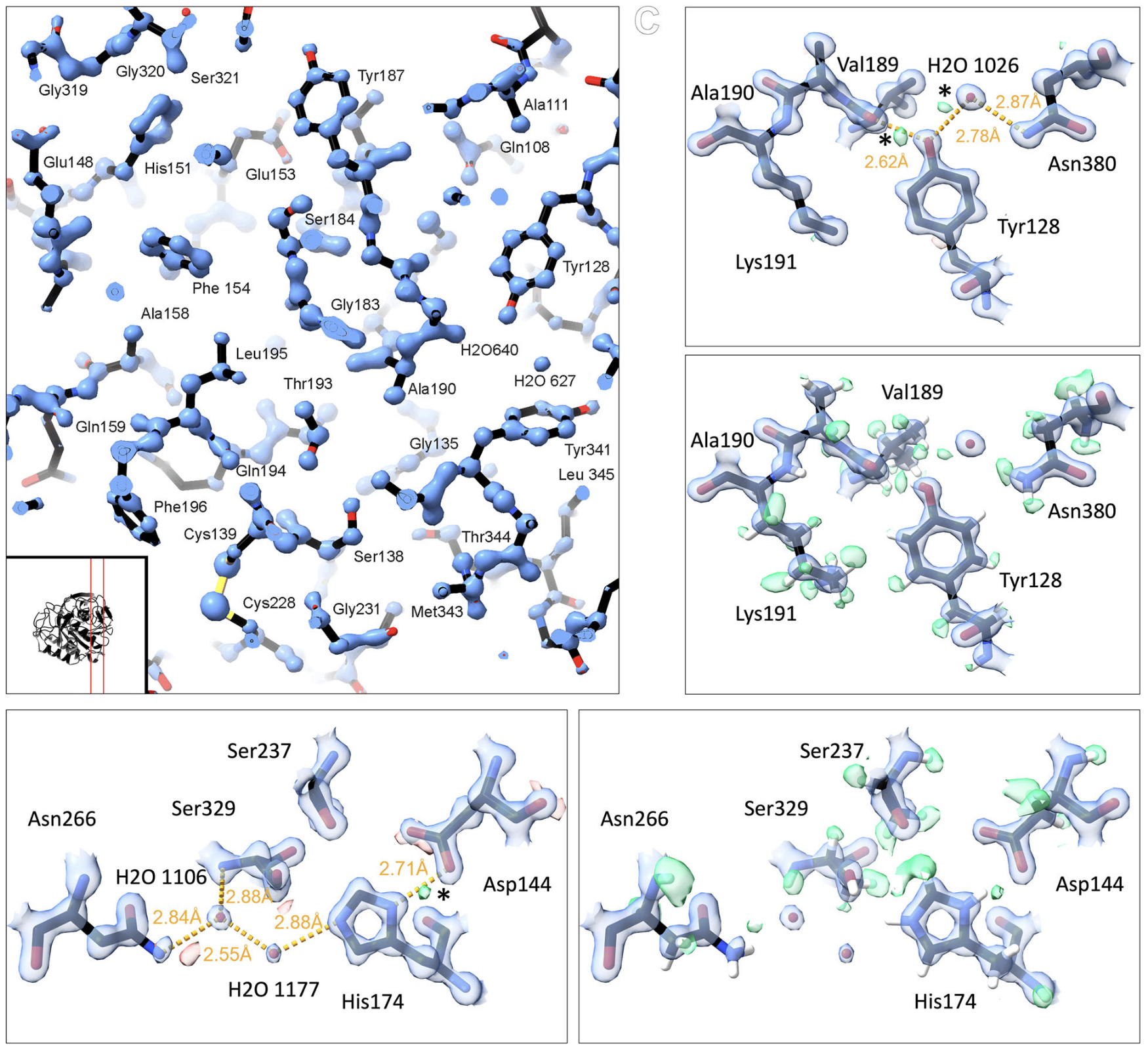
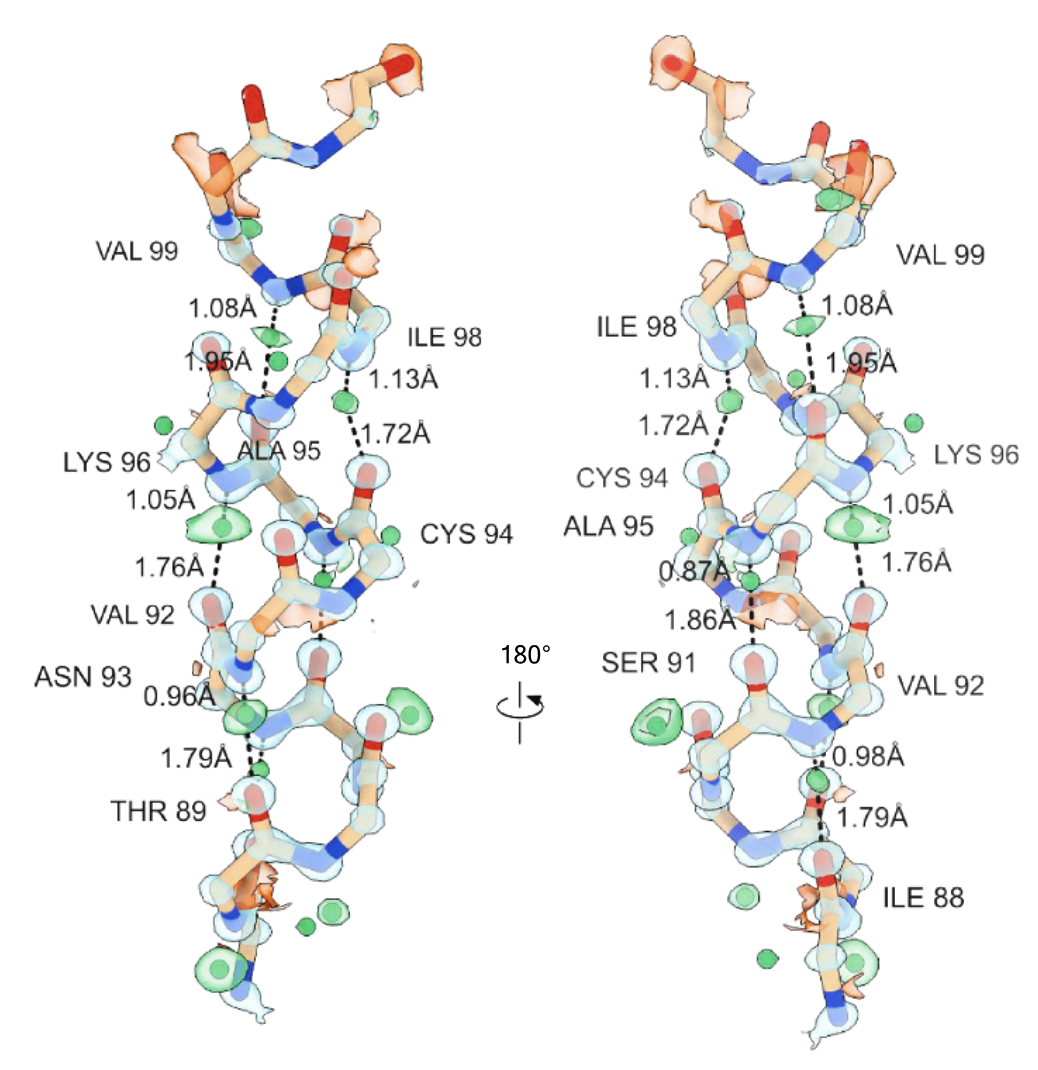
Microcrystal electron diffraction (MicroED) is a powerful technique utilizing electron cryo-microscopy (cryo-EM) for protein structure determination of crystalline samples too small for X-ray crystallography. Electrons interact with the electrostatic potential of the sample, which means that the scattered electrons carry information about the charged state of atoms and provide relatively stronger contrast for visualizing hydrogen atoms. Accurately identifying the positions of hydrogen atoms, and by extension the hydrogen bonding networks, is of importance for understanding protein structure and function, in particular for drug discovery. However, identification of individual hydrogen atom positions typically requires atomic resolution data, and has thus far remained elusive for macromolecular MicroED. Recently, we presented the ab initio structure of triclinic hen egg-white lysozyme at 0.87 Å resolution. The corresponding data were recorded under low exposure conditions using an electron-counting detector from thin crystalline lamellae. Here, using these subatomic resolution MicroED data, we identified over a third of all hydrogen atom positions based on strong difference peaks, and directly visualize hydrogen bonding interactions and the charged states of residues. Furthermore, we find that the hydrogen bond lengths are more accurately described by the inter-nuclei distances than the centers of mass of the corresponding electron clouds. We anticipate that MicroED, coupled with ongoing advances in data collection and refinement, can open further avenues for structural biology by uncovering the hydrogen atoms and hydrogen bonding interactions underlying protein structure and function.
PNAS (2021)
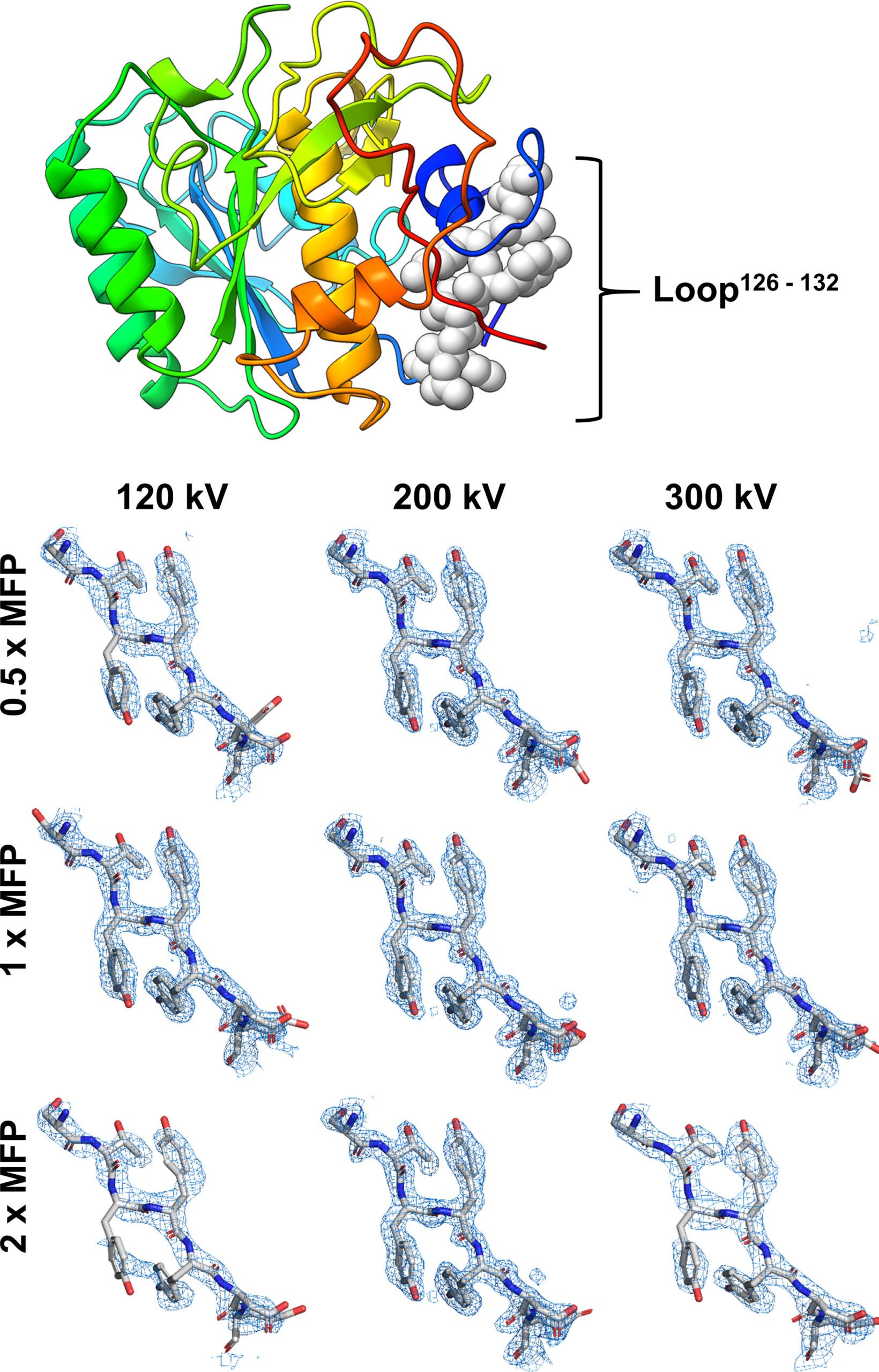
Michael W. Martynowycz, Max T.B. Clabbers, Johan Unge, Johan Hattne, Tamir Gonen
The relationship between sample thickness and quality of data obtained is investigated by microcrystal electron diffraction (MicroED). Several electron microscopy (EM) grids containing proteinase K microcrystals of similar sizes from the same crystallization batch were prepared. Each grid was transferred into a focused ion beam and a scanning electron microscope in which the crystals were then systematically thinned into lamellae between 95- and 1,650-nm thick. MicroED data were collected at either 120-, 200-, or 300-kV accelerating voltages. Lamellae thicknesses were expressed in multiples of the corresponding inelastic mean free path to allow the results from different acceleration voltages to be compared. The quality of the data and subsequently determined structures were assessed using standard crystallographic measures. Structures were reliably determined with similar quality from crystalline lamellae up to twice the inelastic mean free path. Lower resolution diffraction was observed at three times the mean free path for all three accelerating voltages, but the data quality was insufficient to yield structures. Finally, no coherent diffraction was observed from lamellae thicker than four times the calculated inelastic mean free path. This study benchmarks the ideal specimen thickness with implications for all cryo-EM methods.